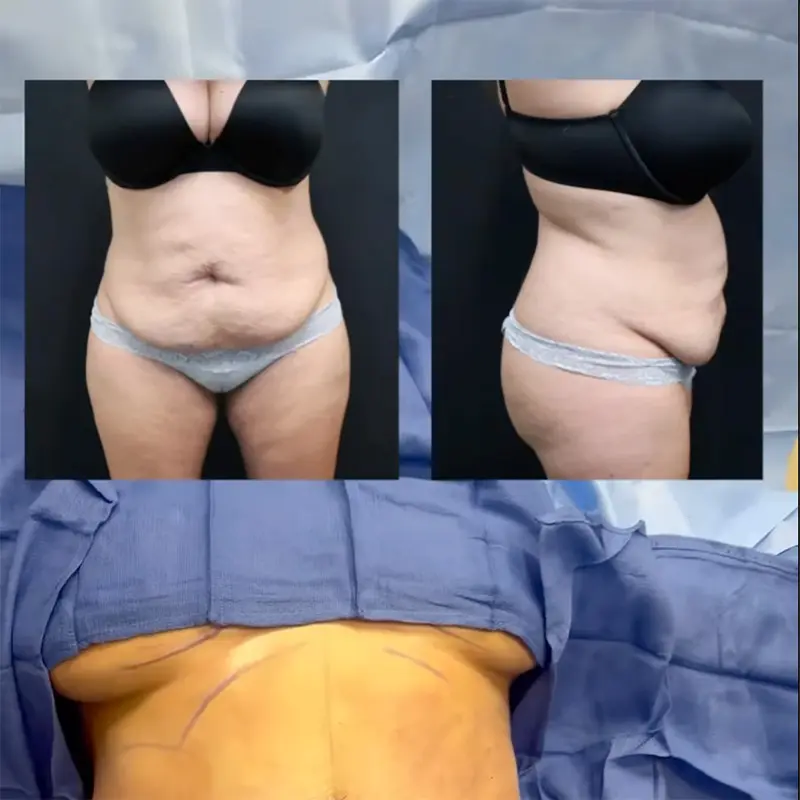
Any surgical procedure carries the risk of complications. Breast augmentation with implants is no different. Breast implants enhance the size, shape, and projection of the breasts for women with naturally small breasts, women whose breasts have flattened with age, weight loss, or pregnancy, or women who have lost their breasts due to mastectomy. When patients choose to undergo breast augmentation, they are often presented with a list of common complications to be aware of, including implant rupture, capsular contracture, changes in skin or nipple sensation, and the possibility of revision surgery. There is, however, another complication that is rarely discussed: breast implant illness. Breast implant illness is not commonly discussed because it is not easily identified. In fact, it is difficult to even prove with complete certainty that implants are causing the adverse physical effects that some women report. So, what is breast implant illness, what should you be on the lookout for, and how can it affect you?
What is Breast Implant Illness?
Hundreds of thousands of women choose breast implants every year. And while most women never experience complications or side effects, there is a select group who have developed similar health problems, and breast implants are the one thing that they have in common. This wide array of symptoms has gradually become known as breast implant illness (BII). Breast implant illness is thought to be an immune reaction to the implant, and therefore develops some of the same symptoms as several autoimmune disorders, which cause the immune system to attack the body. Common symptoms of breast implant illness are thought to include:
- Chronic fatigue
- Mental confusion
- Poor memory
- Joint pain
- Hair loss
- Dry eyes and mouth
- Fevers
- Mood swings
- Shortness of breath
Not every woman with breast implants experiences these adverse effects, and those who do may not see them until years after their surgery.
Are You at Risk?
There has been a growing discussion around the safety of certain breast implants, weighing the risk of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). It’s an issue that has been a priority at the U.S Food and Drug Administration since 2011. As the FDA announced on May 2nd 2019, all breast implant options remain safe and effective and will continue to be available in the U.S. The agency has undertaken several steps to better understand this issue, including an in-depth review of post-approval study data, medical device reports, scientific literature and breast implant-specific registries and public discussions. Importantly panel experts recognized that Board-Certified Plastic Surgeons are the most highly trained to consult, treat and follow up with breast implant patients. We fully support the efforts detailed in the FDA’s announcement and will continue to work with patients to ensure women have the information they need to make informed decisions around breast implants. If you begin to experience any symptoms, call Dr. Mabourakh’s office to set up an appointment and discuss your symptoms in full detail.
For more information about breast implant illness, contact Dr. Mabourakh by calling his Folsom office at (916) 984-8585, his Stockton office at (209) 464-5656, or by filling out our online contact form.